Hajdu Cheney Syndrome: A Detailed Overview
@TCheesy_
1. Genetic Basis:
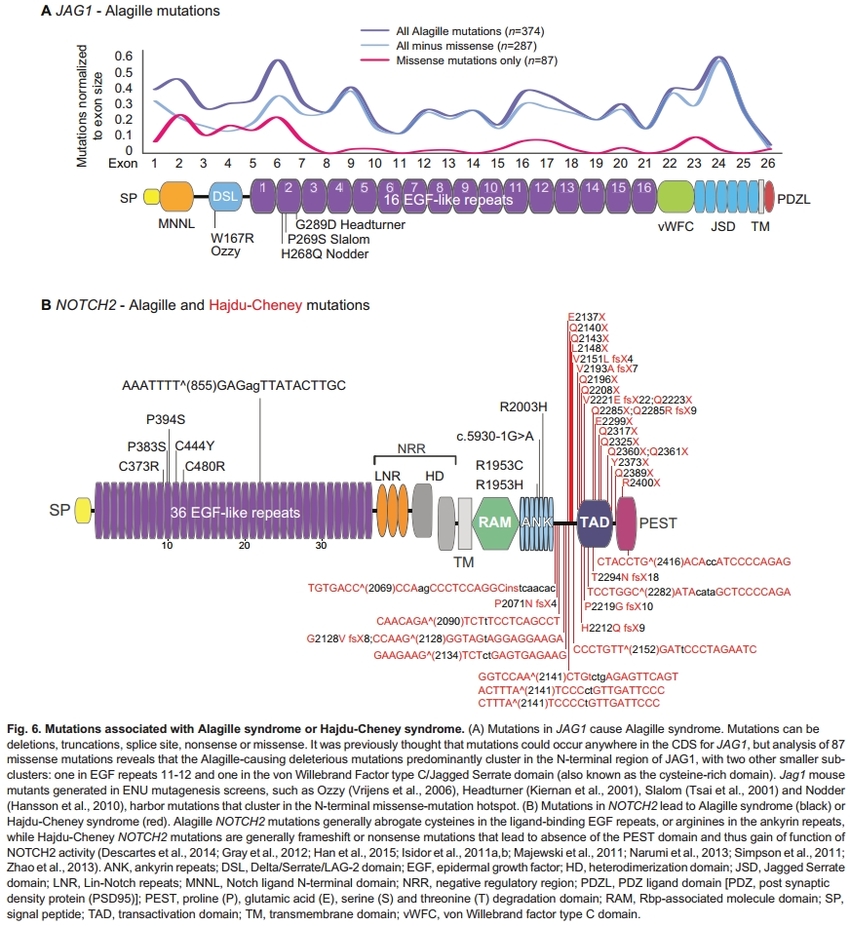
Hajdu-Cheney Syndrome (HCS) is a rare genetic disorder, typically inherited in an autosomal dominant manner, though many cases arise out of nowhere. The condition is primarily linked to mutations in the NOTCH2 gene, which plays a crucial role in the Notch signaling pathway, important for cell differentiation, proliferation, and apoptosis. The mutation in the NOTCH2 gene leads to an abnormal truncation of the protein, affecting the pathway’s function and resulting in the diverse clinical manifestations seen in HCS. I have been tested two times for the genetic errors and none have been found so I remain clinically diagnosed.
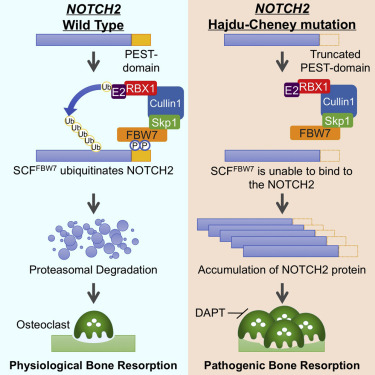
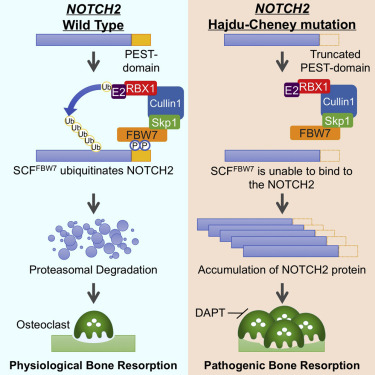
Without Degradation osteoclasts (responsible for dissolving bone) are excessively active and cause osteoporosis through osteolysis.
2. Clinical (physical) Manifestations (expressions):
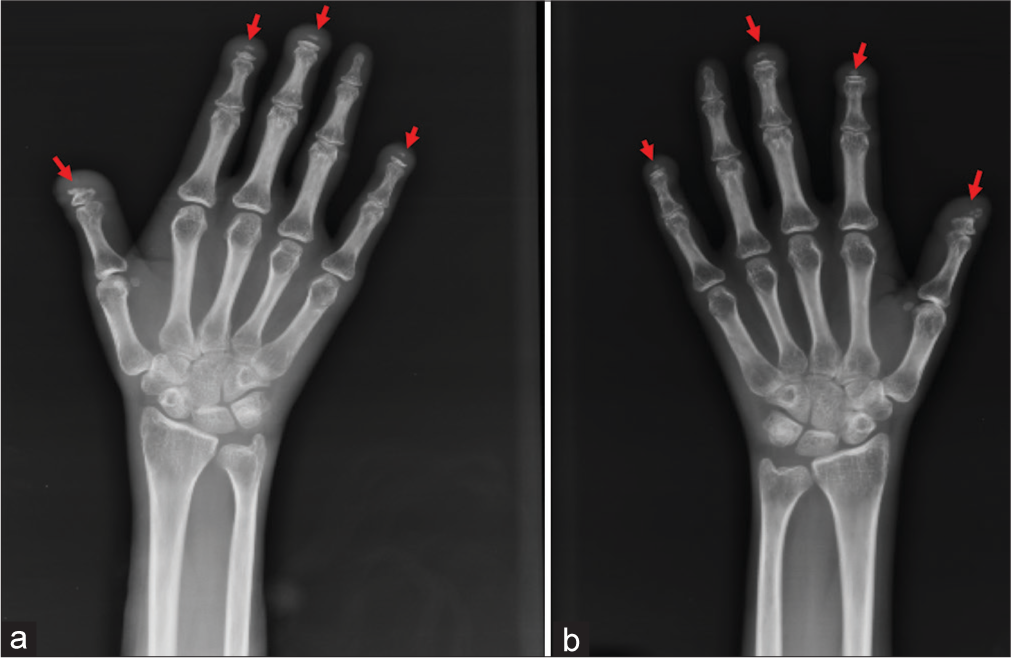
A. Skeletal Abnormalities: HCS is characterized by distinctive skeletal dysplasia. One of the hallmark features is acro-osteolysis—the progressive resorption of the distal (furthest) phalanges (finger bones) , particularly of the fingers and toes. Other skeletal features include:
- Wormian bones: Extra bones within the cranial sutures.
- Osteopenia or osteoporosis: Increased bone fragility, predisposing patients to fractures.
- Spinal anomalies: Including scoliosis, kyphosis, or basilar invagination, which may contribute to neurological complications.
- Premature fusion of the cranial sutures (craniosynostosis): Leading to abnormal skull shape and potentially increased intracranial pressure.
B. Craniofacial Dysmorphisms: Individuals with HCS often exhibit craniofacial abnormalities such as:
- Platybasia: Flattening of the skull base.
- Micrognathia: A small jaw, often leading to dental crowding and malocclusion.
- Midface hypoplasia: Underdevelopment of the central facial bones, contributing to a characteristic facial appearance.
C. Dental Anomalies: Dental issues are prevalent, including:
- Premature loss of teeth.
- Severe periodontal disease.
- Abnormal tooth morphology.
D. Neurological and Developmental Concerns: Neurological symptoms may arise from skeletal abnormalities such as basilar invagination. Cognitive development is typically normal, but some patients may experience delays or learning difficulties secondary to other complications.
E. Cardiovascular Anomalies: Though less common, cardiovascular issues can include:
- Patent ductus arteriosus (PDA).
- Mitral valve prolapse.
- Aortic dilatation or aneurysm.
F. Other Systemic Involvements: HCS may also affect other systems:
- Kidney anomalies: Including renal cysts or nephrocalcinosis.
- Gastrointestinal issues: Such as hernias or other structural abnormalities.
3. Diagnostic Workup:
A. Genetic Testing: Confirmatory diagnosis is typically achieved through genetic testing, identifying mutations in the NOTCH2 gene.
B. Imaging: Radiographic evaluation is critical for assessing the extent of skeletal involvement, including:
- X-rays of the hands and feet to evaluate acro-osteolysis.
- Spinal imaging (X-ray, MRI, or CT) to assess for scoliosis, kyphosis, or basilar invagination.
- Cranial imaging (CT or MRI) to evaluate cranial suture abnormalities or platybasia.
C. Bone Density Measurement: Dual-energy X-ray absorptiometry (DEXA) is often used to monitor bone density, given the risk of osteopenia/osteoporosis.
4. Management and Treatment:
A. Skeletal Management: Managing the skeletal aspects of HCS often involves a multidisciplinary approach:
- Orthopedic interventions for scoliosis or kyphosis.
- Bisphosphonates or other osteoporosis treatments to manage bone density loss, though their efficacy specifically in HCS is not well established.
- Surveillance and early intervention for fractures or other bone complications.
B. Dental Care: Regular dental surveillance is essential, with prompt treatment of periodontal disease and careful monitoring of tooth development.
C. Neurological Management: Patients with basilar invagination or other spinal abnormalities may require:
- Neurological assessment and monitoring for signs of spinal cord compression.
- Surgical intervention in severe cases to decompress the spinal cord or stabilize the cervical spine.
D. Cardiovascular Surveillance: Regular cardiovascular evaluations, including echocardiography, are recommended to monitor for structural heart issues.
E. Genetic Counseling: Given the autosomal dominant inheritance, genetic counseling is crucial for affected families to understand the risks of transmission and to discuss potential prenatal or preimplantation genetic testing options.
5. Prognosis:
The prognosis for individuals with HCS varies widely depending on the severity of symptoms and the extent of systemic involvement. Early diagnosis and a proactive, multidisciplinary approach to management can improve the quality of life and reduce complications. However, the progressive nature of certain aspects, such as acro-osteolysis and osteoporosis, requires ongoing monitoring and management.
Notch2 Signalling
1. Disrupted Cell Differentiation
Overview: The Notch signaling pathway is crucial for guiding the differentiation of cells, which is the process by which a less specialized cell becomes a more specialized cell type. This pathway influences many cell lineages during development, including those in the skeletal system, vascular system, and various organs.
Impact of NOTCH2 Mutation:
- Skeletal Cells (Osteoblasts and Osteoclasts):
- In normal conditions, Notch signaling helps regulate the balance between osteoblasts (cells that form bone) and osteoclasts (cells that resorb bone). A finely tuned balance between these cell types is critical for maintaining healthy bone density and structure.
- In HCS, the mutated NOTCH2 disrupts this balance, leading to abnormal differentiation of osteoclasts. The mutation may cause an overactivation of osteoclasts or a failure to properly regulate their activity. This imbalance leads to excessive bone resorption, which is a hallmark of the syndrome, particularly seen in the acro-osteolysis of the fingers and toes.
- Osteoblast function may also be affected, contributing to insufficient bone formation and further exacerbating bone density loss.
- Chondrocytes (Cartilage Cells):
- Notch signaling also plays a role in the differentiation of chondrocytes, the cells responsible for cartilage formation. Cartilage is critical for the proper development of bones and joints. It is my opinion and experience that this is not given enough consideration when making hospital visits for injuries and pain.
- In HCS, abnormal Notch signaling could result in impaired chondrocyte differentiation, potentially contributing to joint deformities, reduced cartilage integrity, and issues with bone growth and development.
- Craniofacial Development:
- The proper differentiation of craniofacial structures, including the skull and facial bones, is guided by Notch signaling during embryonic development.
- Disruption of this signaling pathway due to the NOTCH2 mutation can lead to the characteristic craniofacial abnormalities seen in HCS, such as macrocephaly (enlarged head), midface hypoplasia, and delayed closure of cranial sutures.
2. Abnormal Bone Remodeling
Overview: Bone remodeling is a continuous process where old bone tissue is removed by osteoclasts and new bone tissue is formed by osteoblasts. This process is essential for maintaining bone strength, repairing micro-damage, and regulating calcium levels.
Impact of NOTCH2 Mutation:
- Osteoclast Overactivity:
- The mutated NOTCH2 gene in HCS leads to an abnormal increase in osteoclast activity. Osteoclasts are responsible for bone resorption, which is the process of breaking down bone tissue and releasing minerals, particularly calcium, into the blood.
- In HCS, this overactivity results in excessive bone loss, particularly in the distal phalanges (finger and toe bones), leading to acro-osteolysis. This bone loss is progressive and can result in shortened, deformed fingers and toes, as well as increased susceptibility to fractures.
- Impaired Bone Formation:
- Not only is bone resorption increased, but bone formation by osteoblasts may also be compromised. The signaling disruption may prevent osteoblasts from functioning optimally, resulting in inadequate bone formation to counterbalance the excessive resorption.
- This imbalance leads to generalized osteopenia (reduced bone density) or osteoporosis (a more severe form of bone density loss), making bones brittle and more prone to fractures even with minor trauma.
- Vertebral and Skeletal Abnormalities:
- The combination of increased osteoclast activity and impaired osteoblast function contributes to the vertebral and skeletal abnormalities observed in HCS patients, such as platyspondyly (flattened vertebrae) and scoliosis (curvature of the spine).
- These structural changes can lead to significant physical disability, chronic pain, and complications related to spinal cord compression or deformities.
3. Vascular Abnormalities
Overview: The Notch signaling pathway is also critical in the development and maintenance of the vascular system, which includes arteries, veins, and capillaries. It plays a role in the differentiation and function of endothelial cells, which line blood vessels, and smooth muscle cells that provide structural support.
Impact of NOTCH2 Mutation:
- Tortuosity of Blood Vessels:
- In HCS, the mutation in NOTCH2 can lead to abnormal vascular development, resulting in tortuosity of blood vessels. Tortuosity refers to blood vessels that are abnormally twisted or have an excessive number of curves.
- This vascular abnormality can affect blood flow and increase the risk of complications such as aneurysms (weakened and bulging areas in the vessel wall) and possibly strokes due to altered or reduced blood flow to critical areas.
- Endothelial Dysfunction:
- Endothelial cells, which line the interior surface of blood vessels, rely on proper Notch signaling for their function and survival. Disruption of this signaling may lead to endothelial dysfunction, a condition where the endothelium (the inner lining of blood vessels) does not function normally.
- This dysfunction can contribute to atherosclerosis (the buildup of fats, cholesterol, and other substances in and on the artery walls), hypertension (high blood pressure), and other vascular diseases, although these specific conditions may not be universally observed in HCS.
- Cardiac Involvement:
- The vascular abnormalities in HCS can also extend to the heart, where Notch signaling is important for the development of the heart’s structural components. The mutation may lead to congenital heart defects, such as septal defects (holes in the heart’s walls), which can affect the heart’s ability to pump blood efficiently.
- Over time, these defects, if not managed, can lead to complications such as heart failure or arrhythmias (irregular heartbeats), impacting the overall health and life expectancy of individuals with HCS.
Summary
In Hajdu-Cheney Syndrome, the mutation in the NOTCH2 gene severely disrupts the Notch signaling pathway, leading to a cascade of problems in cell differentiation, bone remodeling, and vascular development. This disruption manifests in the characteristic symptoms of HCS, such as skeletal deformities, bone loss, and vascular abnormalities, and necessitates careful management to mitigate complications and improve quality of life.
——————————————————————————————————–
The mutation in the NOTCH2 gene associated with Hajdu-Cheney Syndrome (HCS) disrupts several key mechanisms and pathways critical for normal cellular communication and function. Understanding these altered mechanisms and pathways requires a closer look at the normal roles of the Notch signaling pathway and how mutations in NOTCH2 interfere with these processes.
1. Notch Signaling Pathway: Overview
The Notch signaling pathway is a highly conserved cell signaling system present in most multicellular organisms. It regulates cell fate decisions during embryonic development and in adult tissues, controlling processes such as differentiation, proliferation, and apoptosis. The core components of this pathway include:
- Notch Receptors (Notch1-4 in humans): Transmembrane proteins that receive signals from neighboring cells.
- Ligands (Delta-like and Jagged/Serrate family): Transmembrane proteins on adjacent cells that interact with Notch receptors.
- Proteolytic Cleavage: The process that activates the Notch receptor, leading to the release of the Notch intracellular domain (NICD).
- NICD Translocation: The NICD moves to the nucleus and interacts with the CSL (CBF1, Suppressor of Hairless, Lag-1) transcription factor complex, activating the transcription of target genes.
2. Impact of NOTCH2 Mutation on the Notch Pathway
In Hajdu-Cheney Syndrome, the NOTCH2 mutation often results in a truncated or dysfunctional Notch2 receptor, which disrupts the normal signaling cascade. Here’s how these alterations affect the pathway:
A. Impaired Receptor Activation
- Ligand Binding: The mutated Notch2 receptor may have altered or reduced ability to bind its ligands (Delta-like and Jagged/Serrate). This impaired interaction means that the initial signal to activate the pathway is weakened or absent.
- Proteolytic Cleavage: Even if ligand binding occurs, the mutated receptor may not undergo the necessary proteolytic cleavages. Normally, the Notch receptor is cleaved in two steps:
- S2 Cleavage: Occurs at the extracellular side upon ligand binding.
- S3 Cleavage: Occurs at the transmembrane domain, releasing the NICD.
- The mutation in Notch2 may prevent one or both of these cleavages, inhibiting the release of the NICD and thereby blocking downstream signaling.
B. Altered NICD Function
- Nuclear Translocation: In cases where some NICD is still produced, its function may be compromised. The NICD might be less stable, may fail to translocate effectively to the nucleus, or might have reduced ability to interact with the CSL transcription factor complex.
- Target Gene Activation: Even if NICD reaches the nucleus, the interaction with CSL may be impaired, leading to altered or insufficient transcription of Notch target genes. This disruption affects the expression of genes involved in cell differentiation, proliferation, and apoptosis.
3. Downstream Effects on Cellular Mechanisms and Pathways
The malfunctioning Notch2 receptor leads to several downstream consequences:
A. Disrupted Cell Differentiation Pathways
- Skeletal Development (Osteoblasts and Osteoclasts):
- Notch signaling is crucial for the differentiation of mesenchymal stem cells into osteoblasts (bone-forming cells). The Notch2 mutation impairs this process, leading to decreased osteoblast activity and reduced bone formation.
- Simultaneously, the mutation leads to overactivation of osteoclasts, cells responsible for bone resorption. Normally, Notch signaling helps regulate the balance between osteoblasts and osteoclasts, but in HCS, the disrupted signaling results in excessive bone resorption and insufficient bone formation, leading to osteoporosis and acro-osteolysis.
- Neural and Vascular Development:
- In neural tissues, Notch signaling guides the differentiation of neural stem cells into neurons and glial cells. The mutation may lead to abnormal neural differentiation, contributing to cognitive and developmental delays.
- In the vascular system, endothelial cell differentiation and angiogenesis (formation of new blood vessels) are influenced by Notch signaling. The mutation can lead to abnormal vessel formation, resulting in vascular tortuosity and potential cardiovascular complications.
B. Dysregulation of Apoptosis
- Programmed Cell Death: Notch signaling can promote cell survival in some contexts and induce apoptosis in others, depending on the cell type and developmental stage. The mutation in NOTCH2 may lead to inappropriate activation or suppression of apoptosis:
- Excessive Cell Death: In certain tissues, the disrupted signaling could lead to increased apoptosis, contributing to tissue degeneration and the characteristic bone and connective tissue abnormalities in HCS.
- Inhibited Cell Death: Conversely, in other tissues, the mutation may prevent normal apoptosis, leading to the survival of abnormal cells and contributing to pathological tissue remodeling.
C. Aberrant Gene Expression
- Target Genes: Notch signaling regulates a wide array of target genes, including those involved in cell cycle regulation, differentiation, and metabolic processes. The mutation in Notch2 can result in the following:
- Reduced Expression: Critical genes required for normal development and tissue maintenance may be under-expressed, leading to developmental defects and tissue degeneration.
- Misexpression: In some cases, abnormal gene expression might occur, where genes are turned on or off inappropriately, leading to pathological changes in tissue structure and function.
4. Broader Systemic Impacts
The disrupted Notch signaling due to the NOTCH2 mutation does not only affect specific cell types but has broader systemic implications:
A. Bone Remodeling
- Systemic Osteopenia/Osteoporosis: The imbalance between osteoclast and osteoblast activity leads to systemic bone loss, which is a major feature of HCS. This makes bones more susceptible to fractures and deformities.
B. Connective Tissue Integrity
- Joint and Craniofacial Abnormalities: The mutation affects the development and maintenance of connective tissues, leading to joint laxity, craniofacial deformities, and issues with the integrity of the skeletal system overall.
C. Cardiovascular System
- Vascular Tortuosity and Aneurysms: The altered signaling pathway leads to abnormal development of the vascular system, increasing the risk of vessel abnormalities, which can cause complications such as aneurysms or hemorrhages.
5. Potential Therapeutic Implications
Understanding these disrupted pathways provides insights into potential therapeutic strategies:
- Gene Therapy: Correcting or compensating for the mutated NOTCH2 gene could potentially restore normal signaling.
- Targeted Molecular Therapies: Drugs that modulate Notch signaling or specifically target the downstream effects of the mutation might help mitigate some of the pathological features of HCS.
- Bone-Targeted Therapies: Agents that inhibit osteoclast activity or promote osteoblast function could help restore bone density and prevent fractures.
Summary
The mutation in NOTCH2 leads to a cascade of disruptions in the Notch signaling pathway, affecting critical processes like cell differentiation, apoptosis, and gene expression. These disruptions manifest in the characteristic features of Hajdu-Cheney Syndrome, including severe bone loss, vascular abnormalities, and connective tissue disorders. Understanding these altered mechanisms is crucial for developing targeted therapies that could alleviate the symptoms or slow the progression of this debilitating condition.
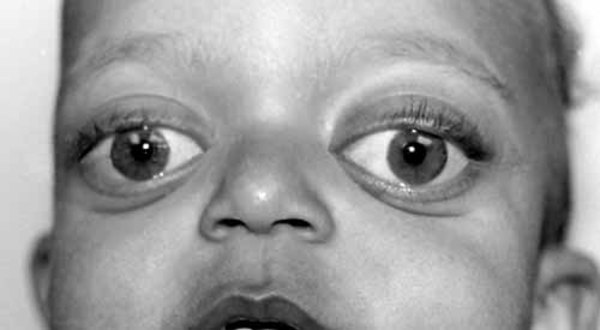
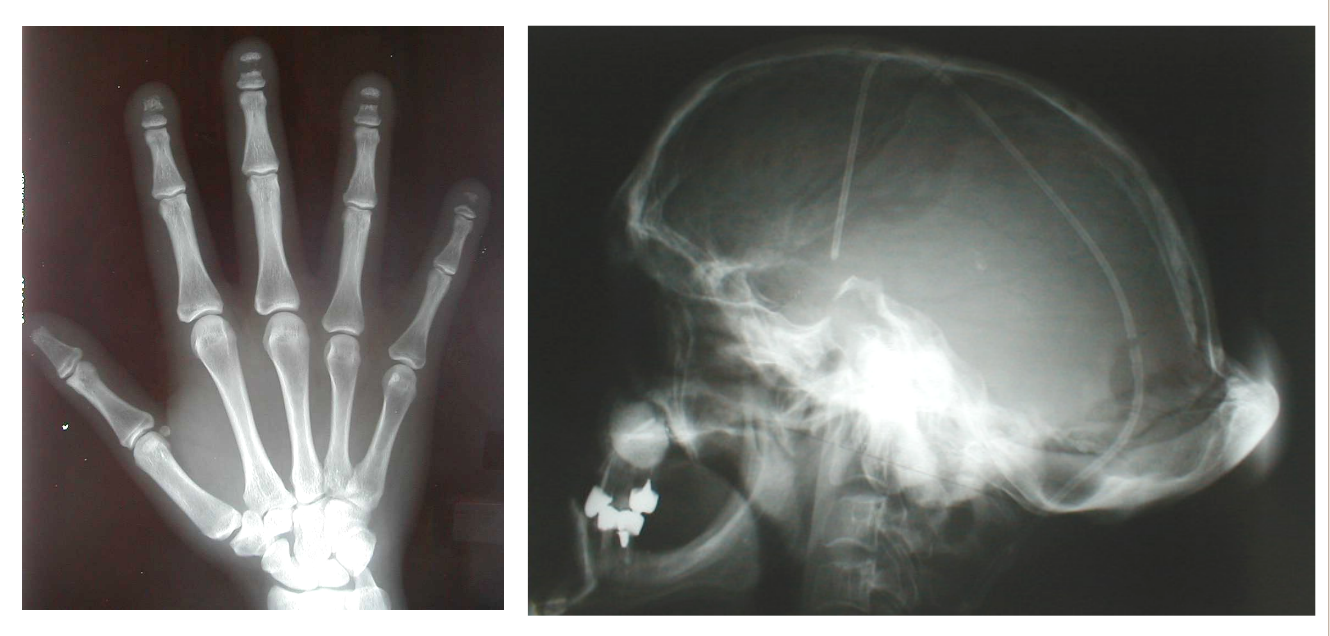
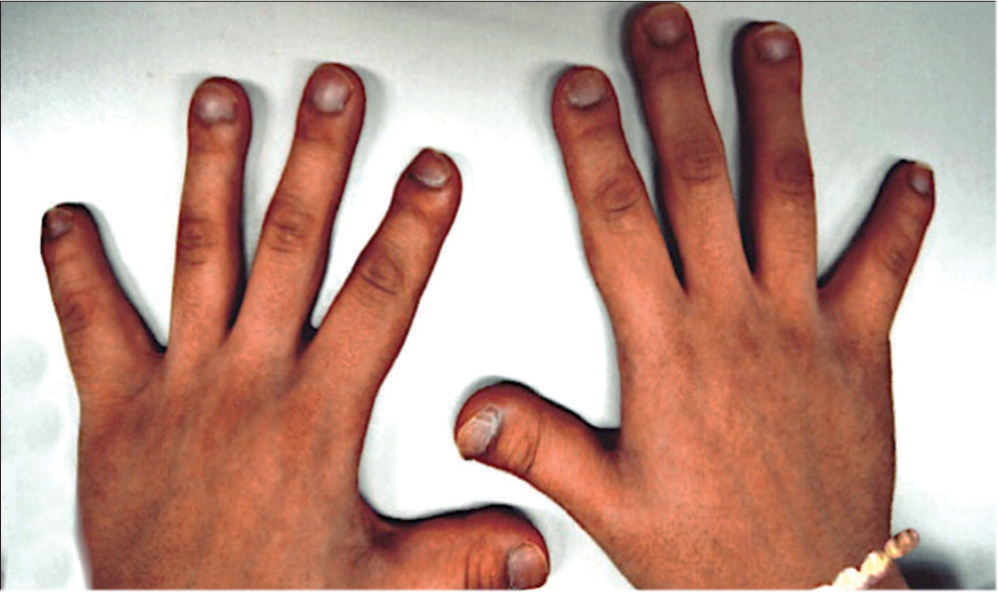
Hajdu Cheney Syndrome is varied in all of us who have it so I feel it’s important to be specific about how it is expressed in me today, yesterday and through my past.


Leave a word